ToF SIMS – Time of Flight Secondary Ion Mass Spectrometry
What is ToF SIMS? What is it used for, and what sort of information can it provide? Which samples are suitable (and which are not)? In this series, we will answer all these questions and more.
Time-of-Flight Secondary Ion Mass Spectrometry (ToF SIMS) is a surface analysis technique used to study the chemical composition of solid surfaces and thin films in three dimensions.
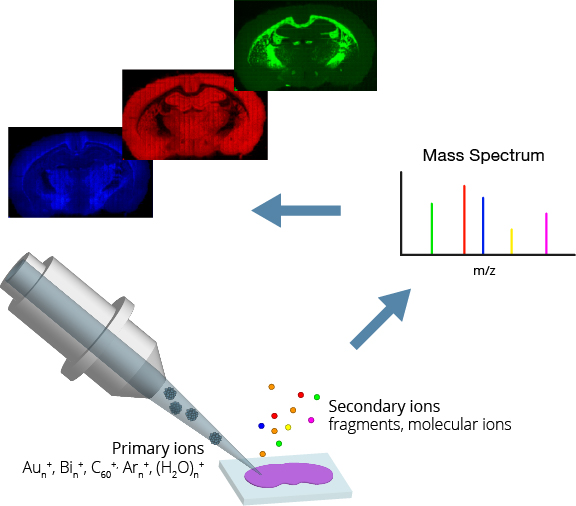
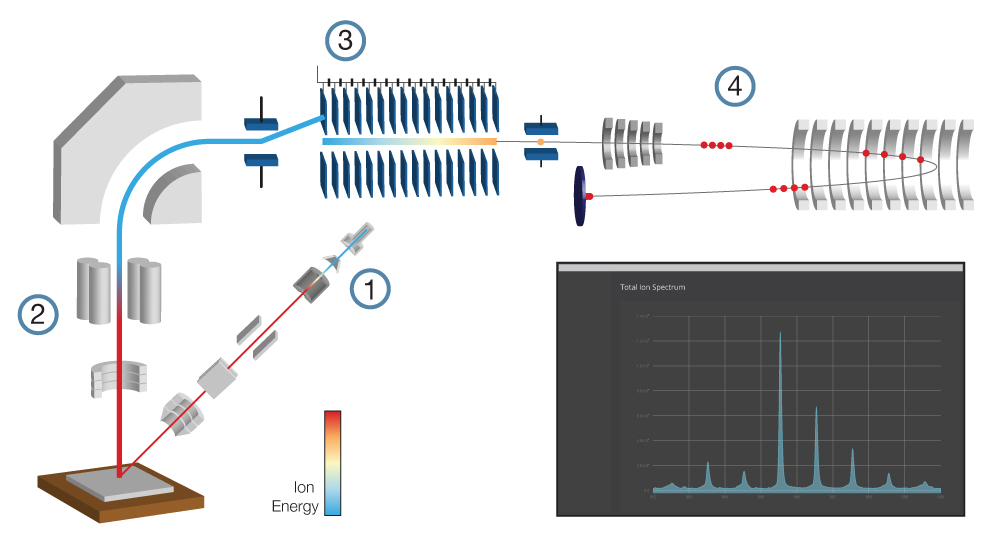
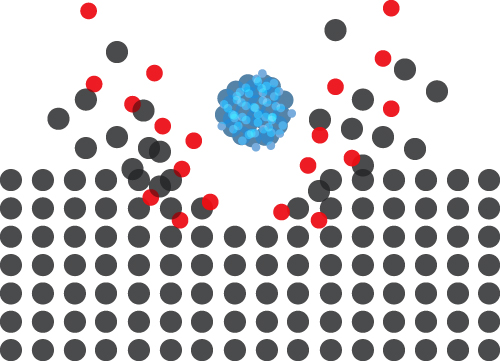
A focused beam of primary ions bombards a target surface, creating a plume of neutral atoms/molecules, secondary ions, and electrons. The secondary ions are collected and analysed using a time-of-flight mass spectrometer. The mass spectrometer measures an ion’s mass-to-charge ratio (m/z) by precisely timing how long it takes to reach the detector – the “time of flight”.
By scanning the primary ion beam across an area of the sample, a chemical map of the surface is formed pixel by pixel. Scientists and technicians use ToF SIMS daily for fundamental research, routine analysis, and quality control in academic and industrial settings.
For many years, the limitations of the primary ion beam confined the analysis to looking at atomic species and small molecules. With advances in instrument and ion beam design, modern instruments such as the J105 SIMS are now routinely imaging large intact molecules. These new capabilities have caused an explosion in new applications, and more papers are published each year in bio and bio-related fields using ToF SIMS.
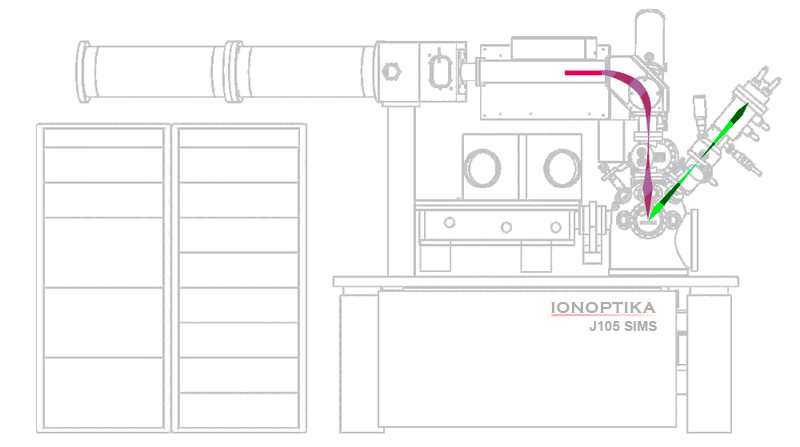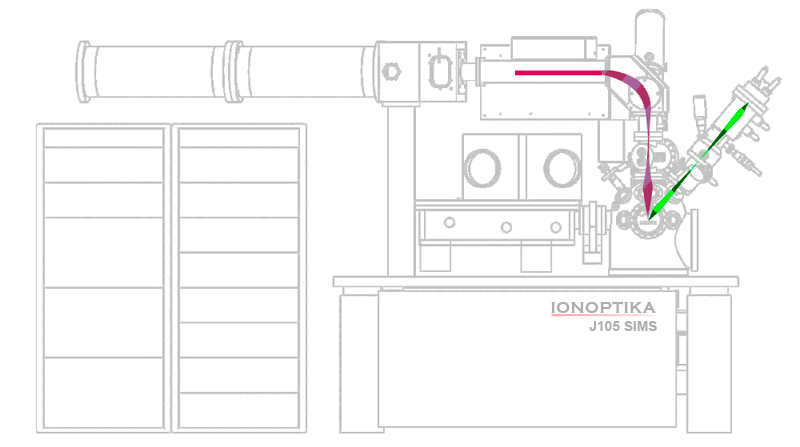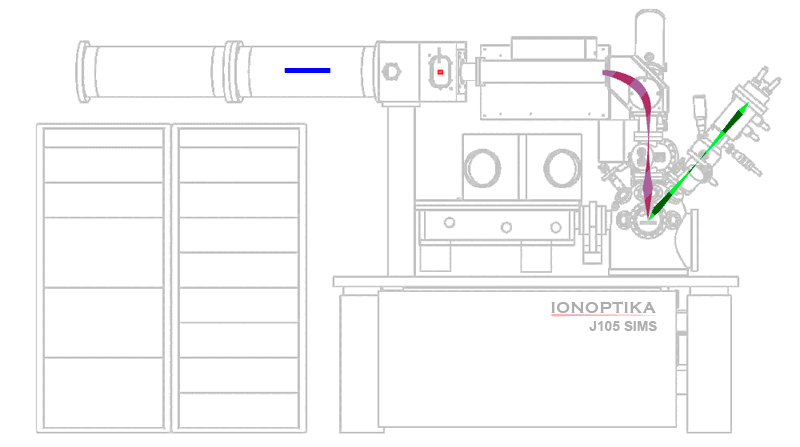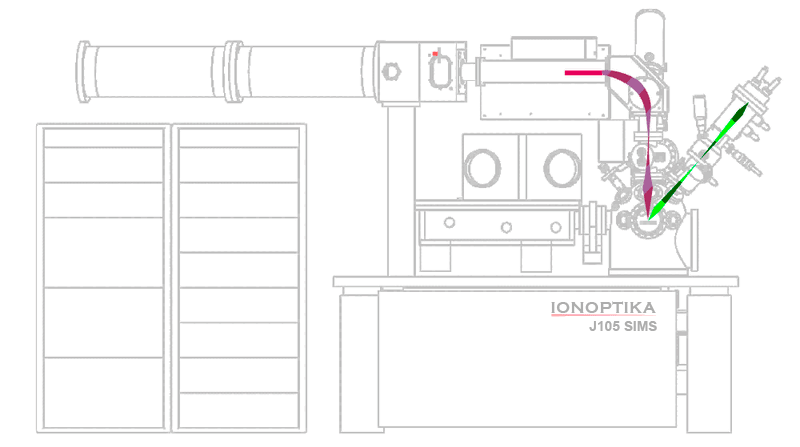
Anatomy of a ToF SIMS instrument
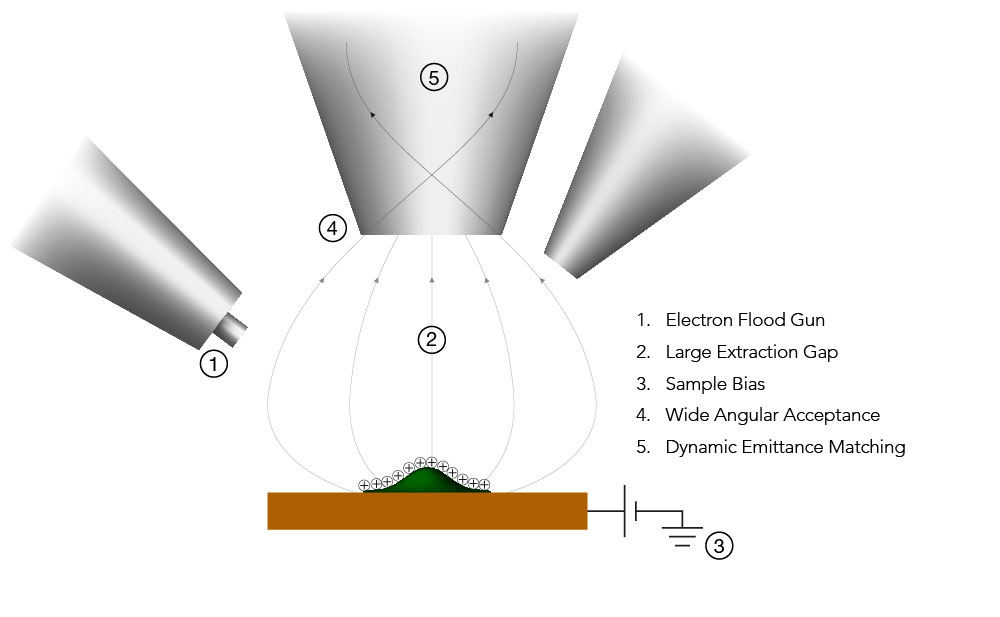
ToF SIMS instruments are often larger and more expensive than most other analytical instruments found in a lab. High-vacuum conditions (< 1×10-6 mbar) are required to prevent ions from colliding with gas molecules in the air, requiring bigger vacuum pumps, more robust seals, and additional precautions to prevent leaks.
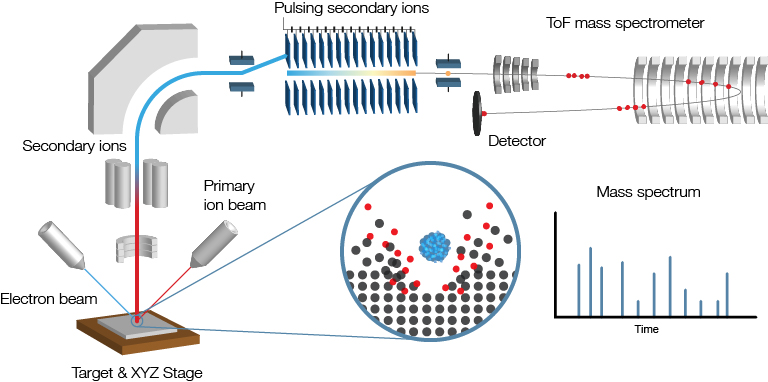
| Primary components | Secondary components |
|---|---|
| Sample analysis chamber (SAC) | Sample introduction System |
| Primary ion beam | Cryogenic cooling for low-temperature analysis |
| Secondary ion extraction optics | Charge compensation, e.g., electron beam |
| Mass spectrometer | Secondary electron imaging |
Key Benefits of ToF SIMS
- Spatial resolution. ToF SIMS achieves significantly higher spatial resolutions than other imaging methods, thanks to beam sizes as small as a few hundred nanometres.
- Speed. The time-of-flight mass spectrometer operates at much higher rates than other MS techniques. ToF SIMS instruments can run at speeds up to 1000 pixels per second.
- 3D imaging. The primary ion beam removes a small amount of material each time it scans across the surface. By making multiple passes over the same area, a 3D map of the material builds up layer by layer.
- Sensitivity. Small spot sizes and shallow impact craters result in tiny analysis volumes, which require great care to prevent signal loss. As a result, SIMS is generally more sensitive than other forms of mass spectrometry.
- Dynamic range. The ions in a ToF SIMS spectrum can range from a single hydrogen ion to intact protein molecules several thousand daltons in size.
- Applications. The breadth of applications for ToF SIMS is enormous, ranging from metallurgy to fundamental biology and most things in-between.
Applications of ToF SIMS
ToF SIMS provides a detailed three-dimensional chemical map of a sample. Information about the atoms and molecules that make up the sample, their distribution, and any contamination present are all revealed. This type of information is beneficial for many applications.
Academic research labs, industrial quality control, and research organisations use ToF SIMS daily. Disciplines as diverse as materials science, analytical chemistry, biology, geology, pharmaceutical science, and many others benefit from the detailed chemical information ToF SIMS provides.
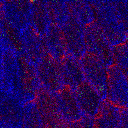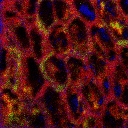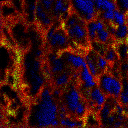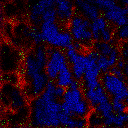
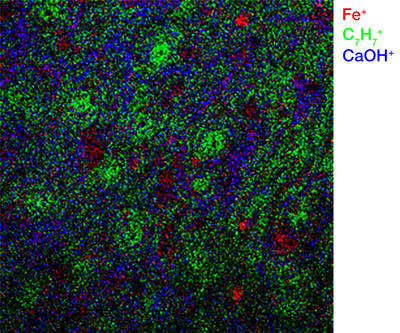
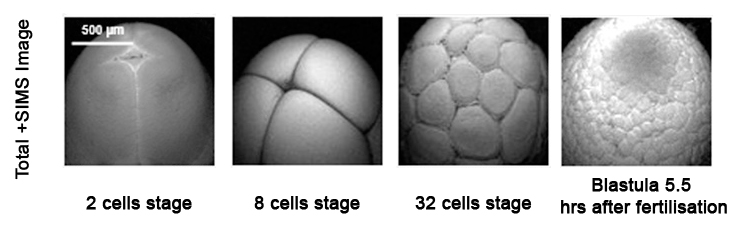
2D Imaging
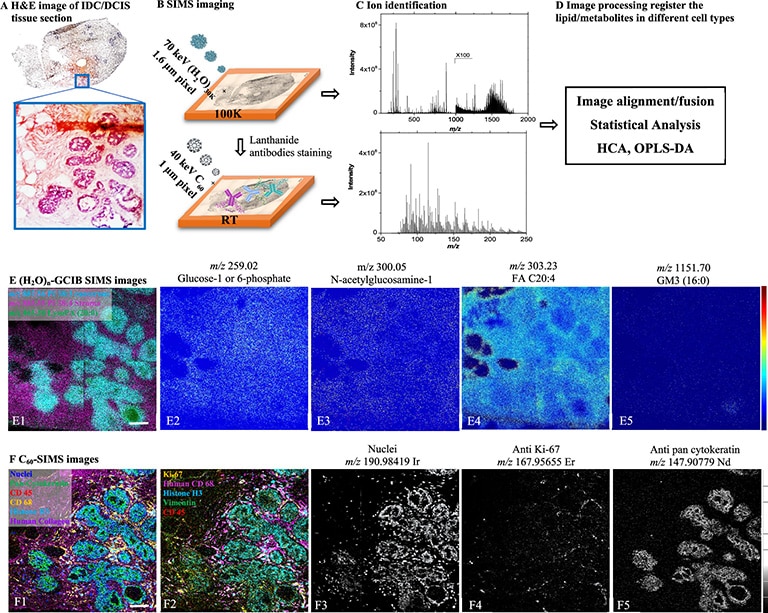
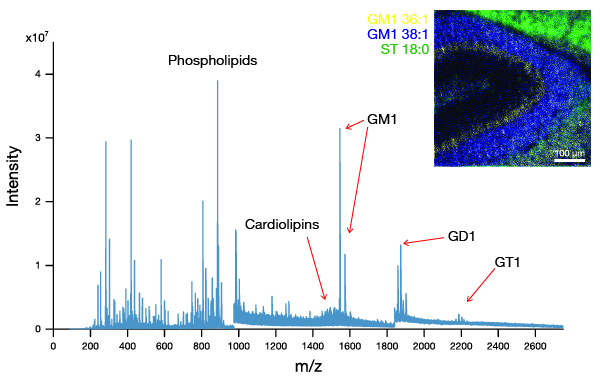
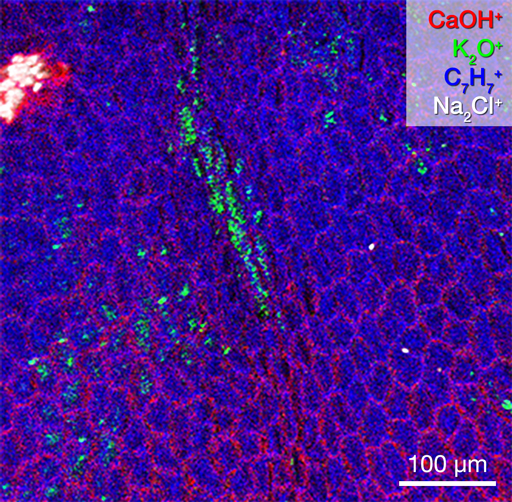
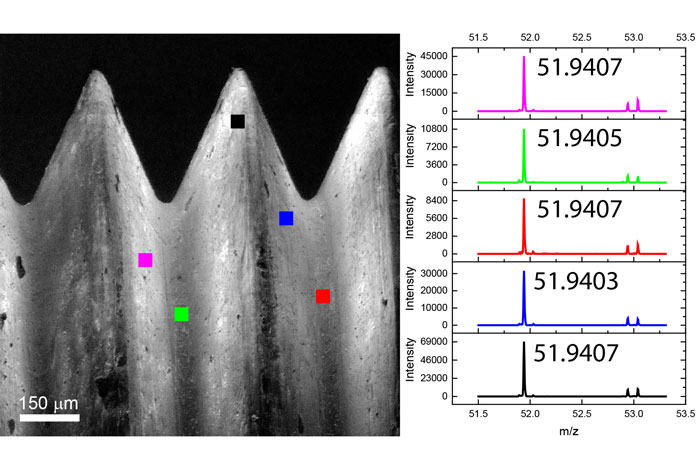
2D images are the most common mode of operation for ToF SIMS applications, whereby the ion beam scans the surface, acquiring a mass spectrum at each pixel. The image resolution can vary from a few hundred pixels to over four million.
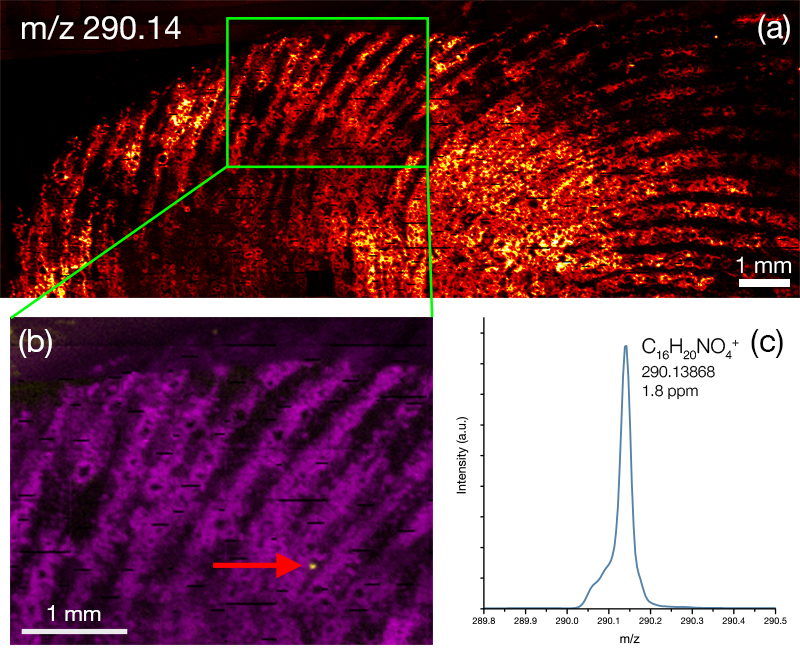
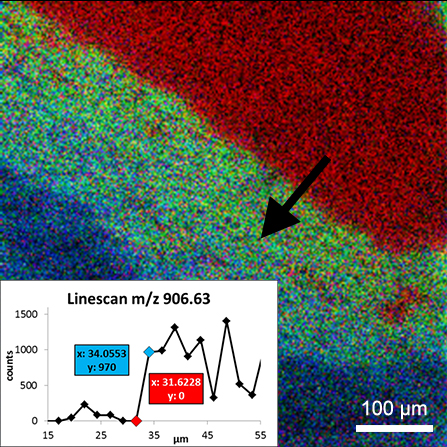
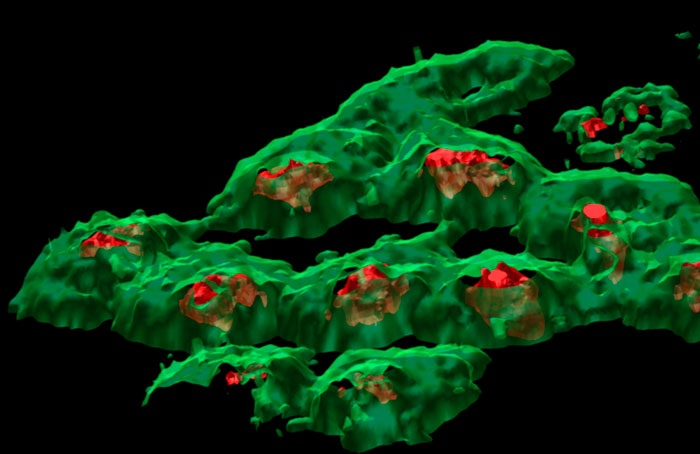
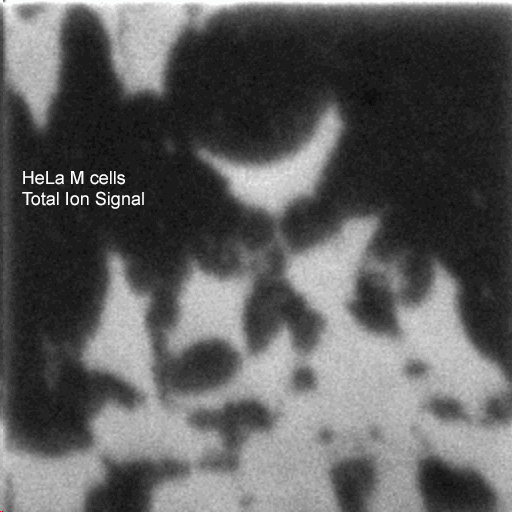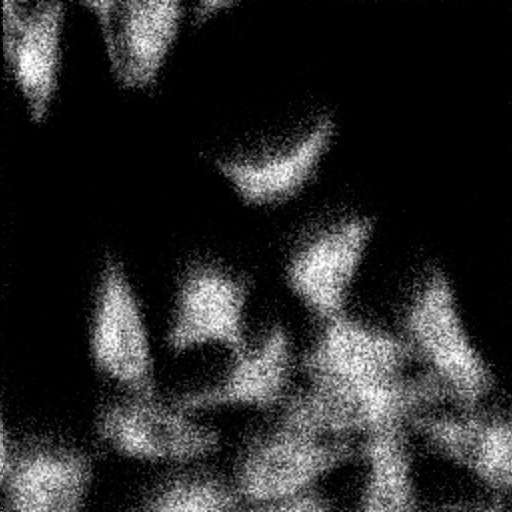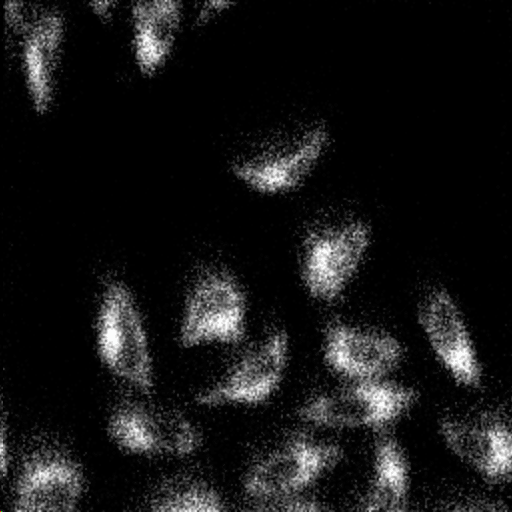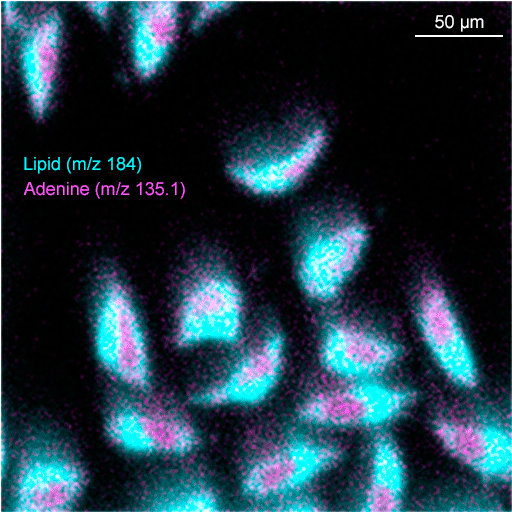
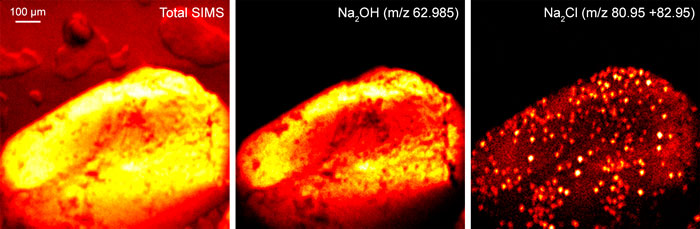
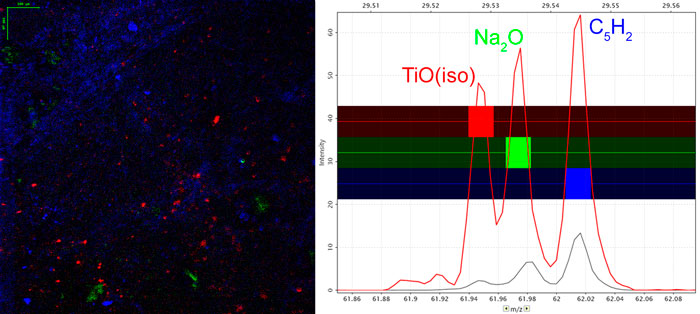
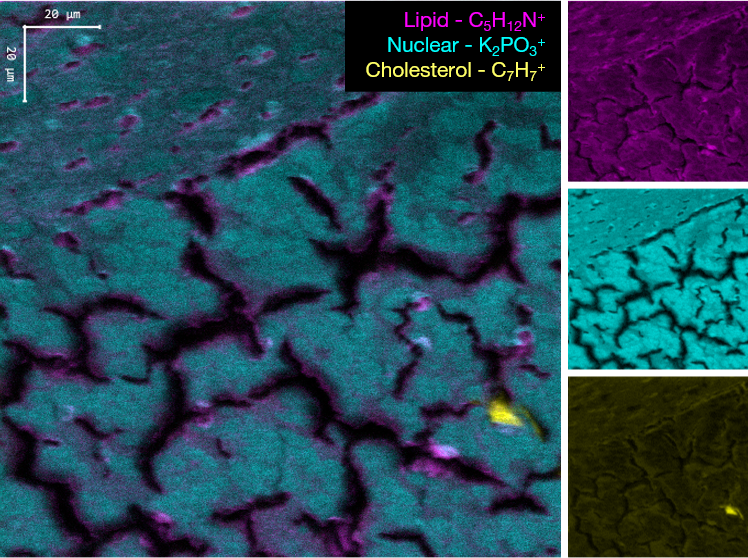
Images of individual mass channels show the precise distribution of ions across the field of view. Overlaying multiple mass channels can show the distribution of different ions and how they relate to each other.
The image below shows three individual ion images and an overlay image representing different components of a biological tissue sample.
Spectrometry
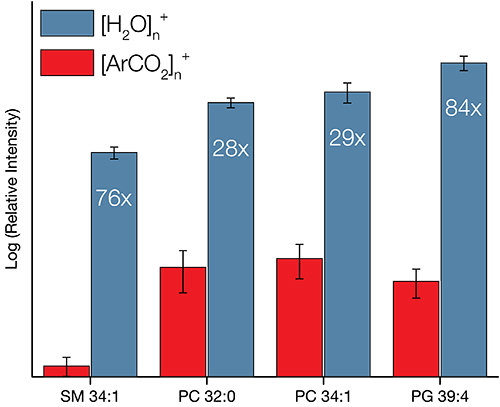
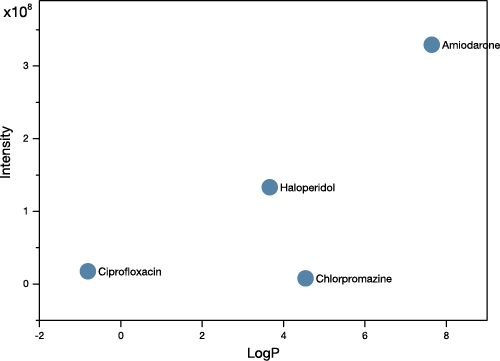
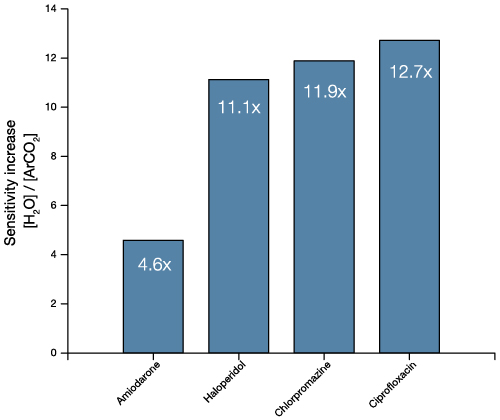
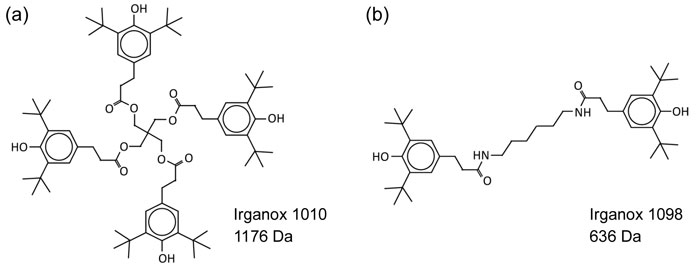
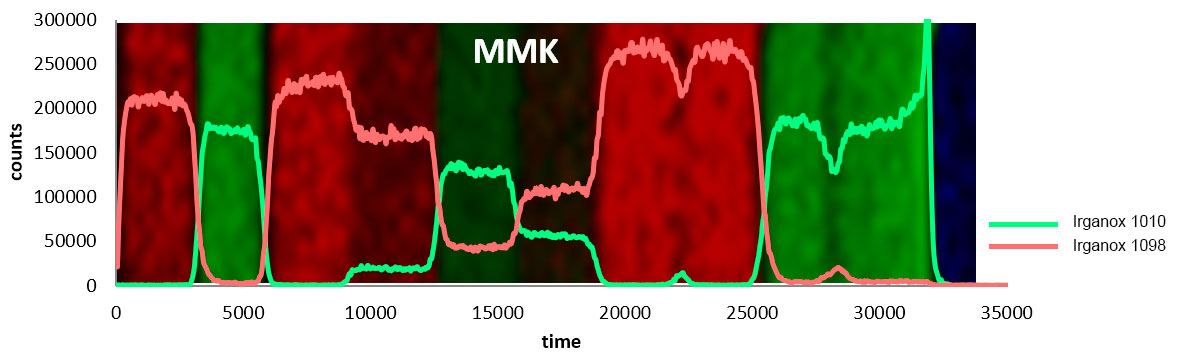
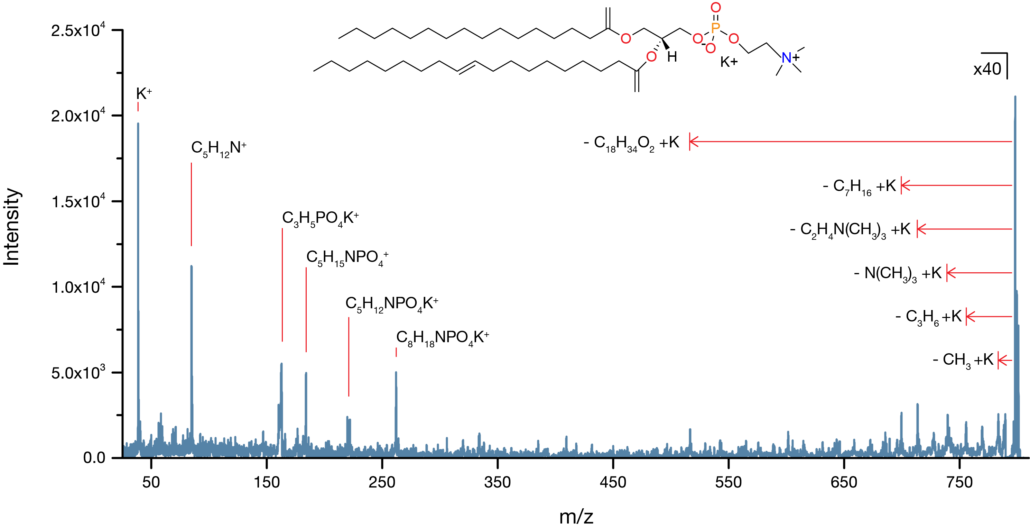
Analysis of a ToF SIMS spectrum provides information on the atomic or molecular makeup of the sample and can inform about the general abundance of various compounds. It is also possible to determine atomic ratios in some cases, but this requires well-controlled samples and careful use of reference materials.
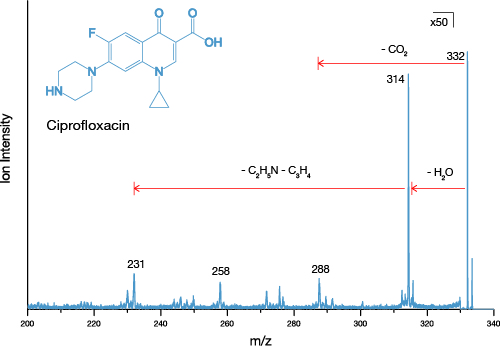
Tandem mass spectrometry is a feature on most major ToF SIMS instruments and is extremely useful for confidently identifying ions. Tandem MS, also known as MS/MS, or MS2, involves isolating a secondary ion of interest, fragmenting it, and collecting the resulting fragments in a mass spectrum. By analysing the daughter peaks, it is possible to determine the parent ion with a high level of precision.
Depth profiling
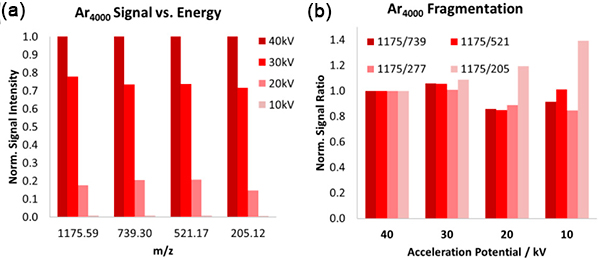
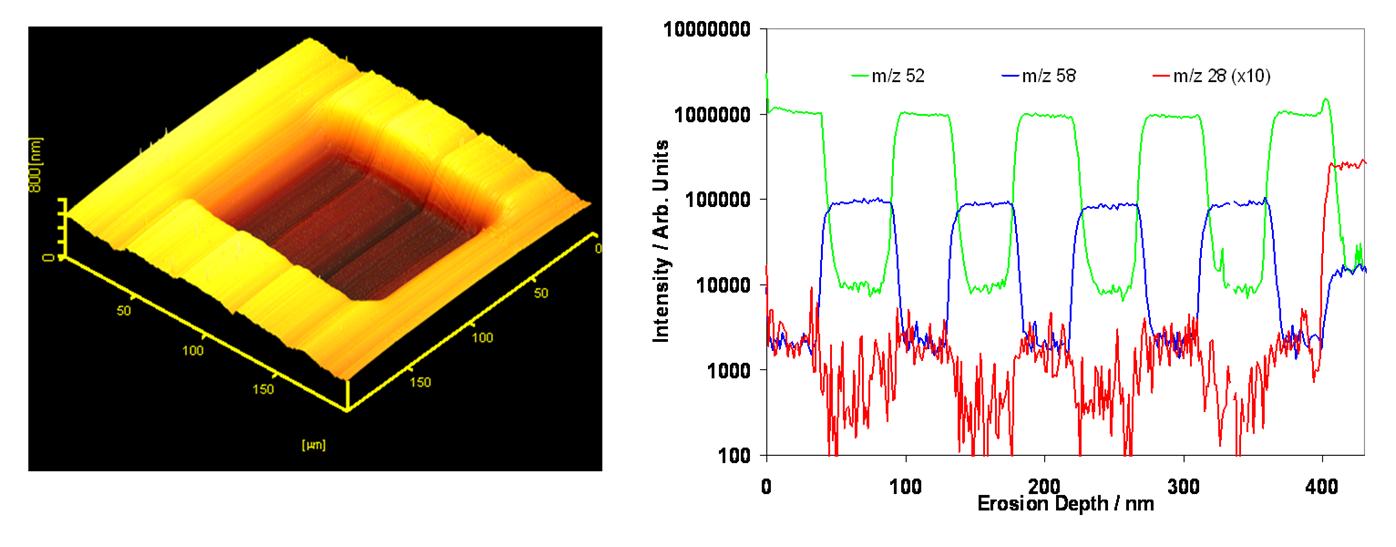
A powerful analysis mode, depth profiling involves etching vertically through the sample and acquiring a mass spectrum at every layer. The result is a profile of all atoms/molecules through the sampled volume. Large cluster ions reduce damage to sub-surface layers, minimising interlayer mixing and maximising depth resolution. With the right ion beam and sample combination, depth resolution as low as a few nanometres is possible.
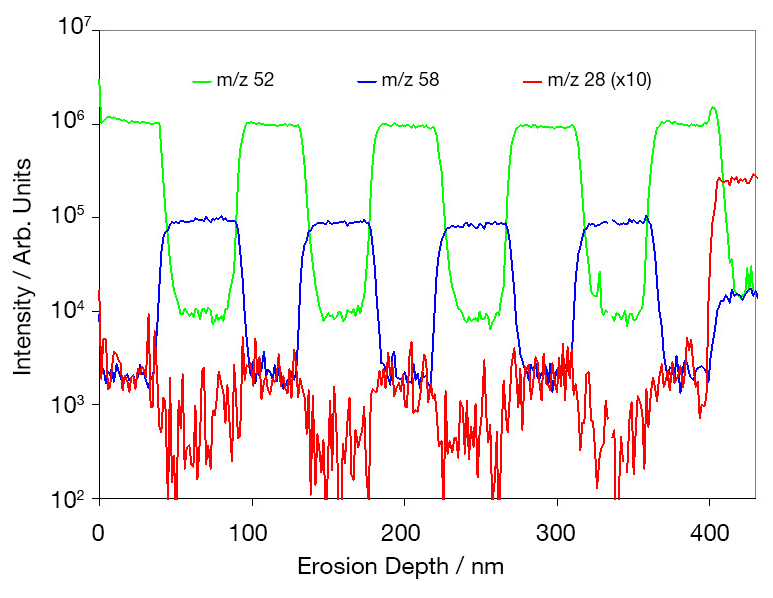
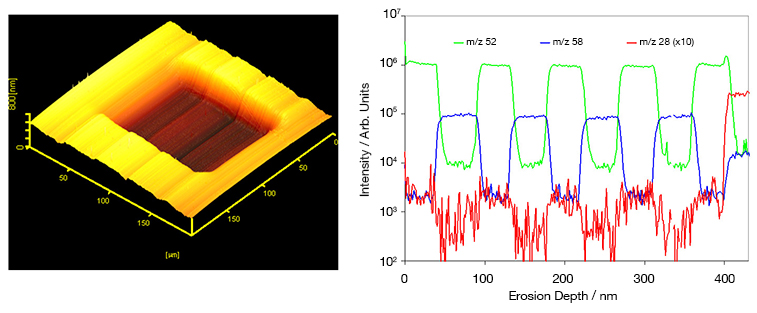
Depth profile through the NIST Ni/Cr standard reference material using a C60 beam, showing 5 nm depth resolution.
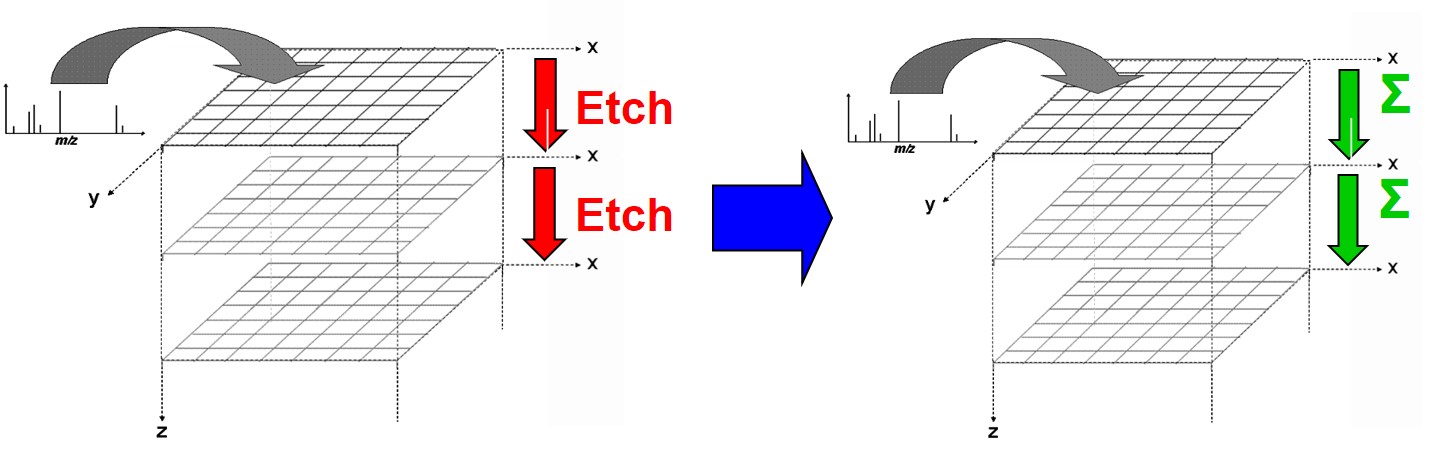
3D Imaging
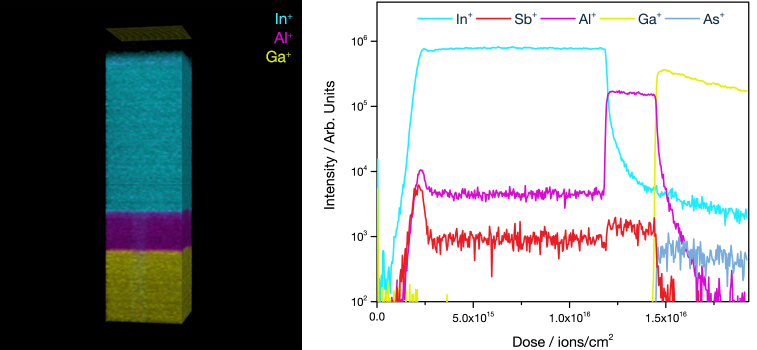
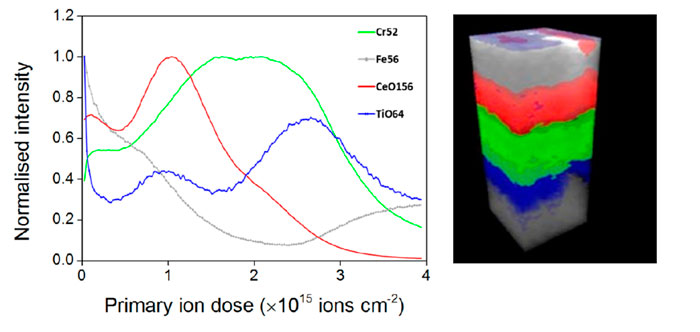
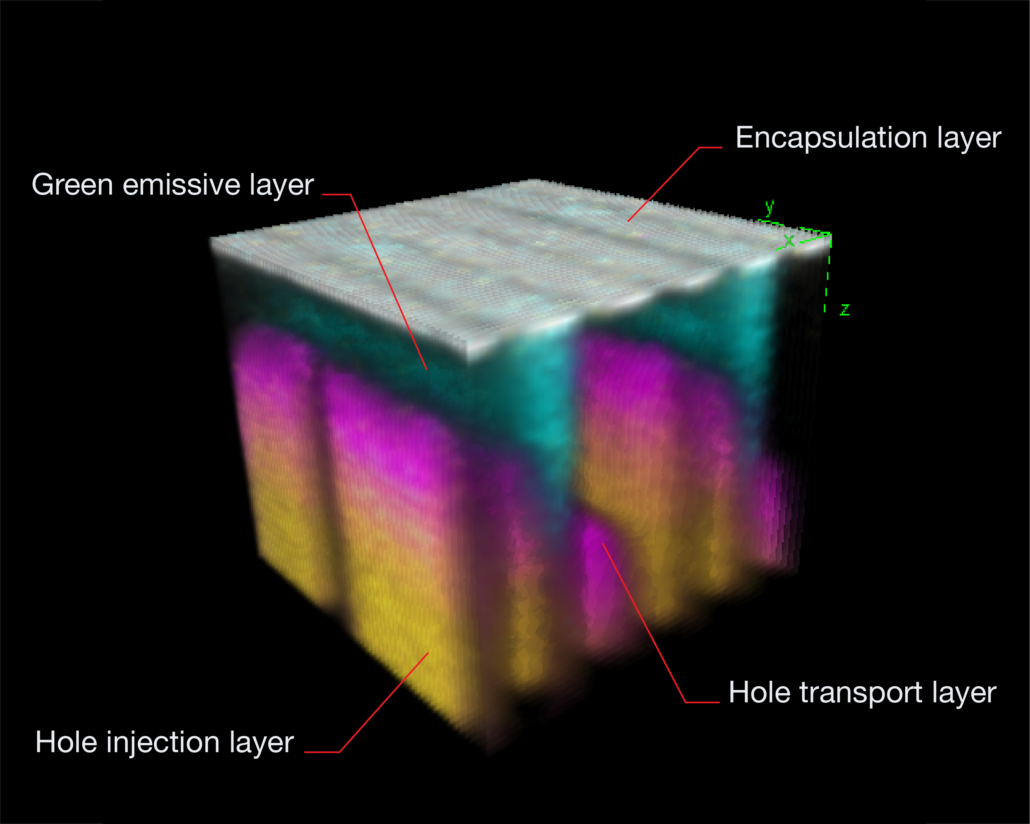
The feature that sets ToF SIMS apart from other mass spectrometry and analytical techniques is the ability to acquire 3D data sets. Like a depth profile, a 3D analysis involves acquiring many 2D layers repeatedly over the same area, etching material with each pass, and building up a three-dimensional view of the sample. Large cluster ions are ideal for 3D analysis as they produce very little damage and can therefore be used to etch and analyse the sample simultaneously.
Unlike techniques like AFM, which capture the 3D topography of the sample, SIMS cannot distinguish 3D objects from flat objects. The technique works best for flat samples with layers of interest below the surface, as in the OLED example below. It is possible to reconstruct the topography of a non-flat sample; however, this requires prior knowledge of the material structure.
Read more about the applications of ToF SIMS in our Application Notes section. Or, to dive deeper into more advanced topics, check out the list of publications using our equipment here. You might also like to learn more about how the J105 SIMS operates, which you can read here.